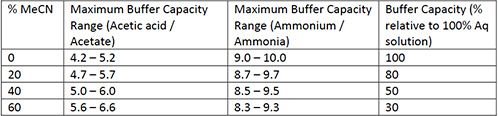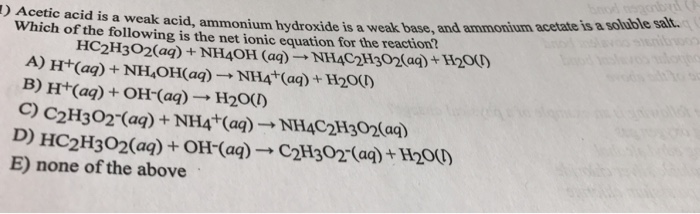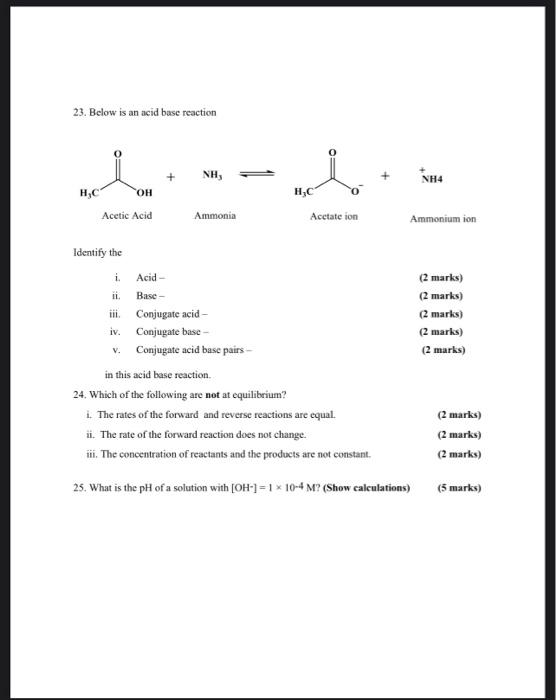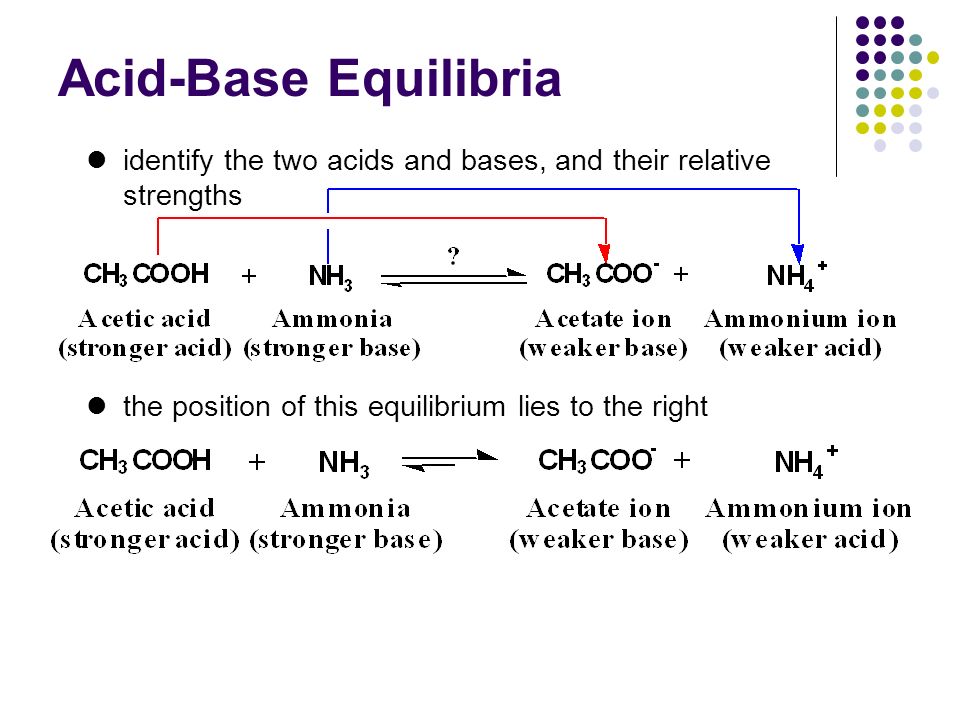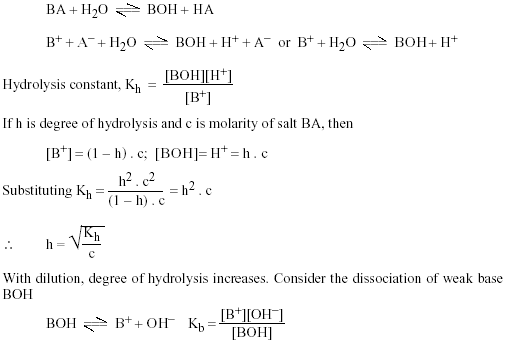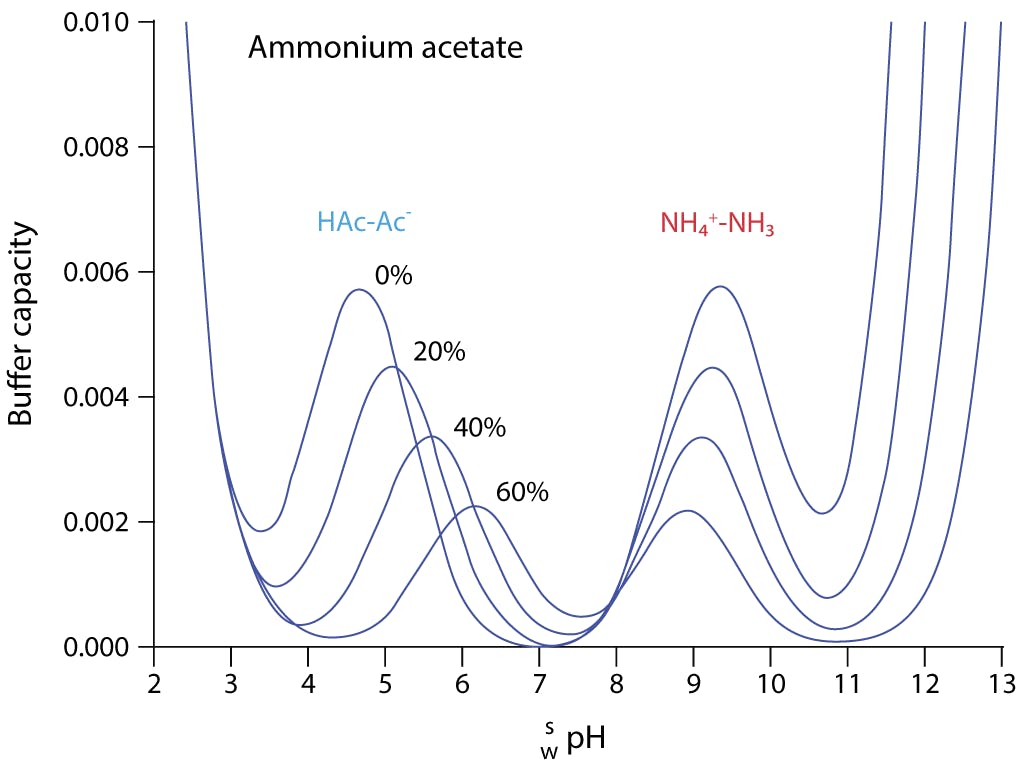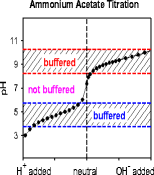
Addressing a Common Misconception: Ammonium Acetate as Neutral pH “Buffer” for Native Electrospray Mass Spectrometry | SpringerLink
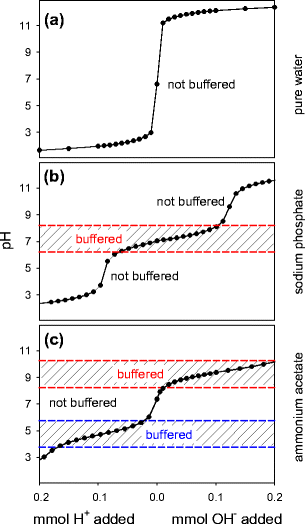
Addressing a Common Misconception: Ammonium Acetate as Neutral pH “Buffer” for Native Electrospray Mass Spectrometry | SpringerLink

Ammonium acetate (C<sub>2</sub>H<sub>7</sub>NO<sub>2</sub>) - Structure, properties , Production, Uses and FAQs of Ammonium acetate.

science chemistry acid base indicator bromothymol blue | Fundamental Photographs - The Art of Science

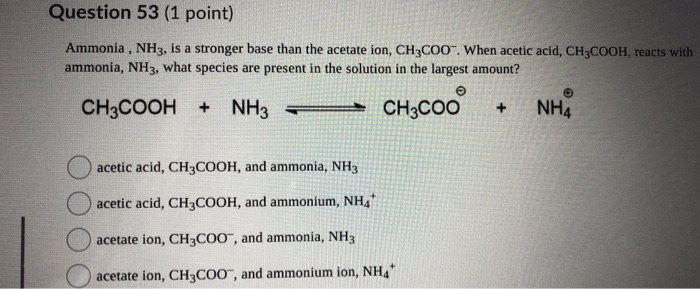
pka of acetic acid and pKb of ammonium hydroxide are 4.76 and 4.75 respectively. Calculate the pH of ammonium acetate solution.