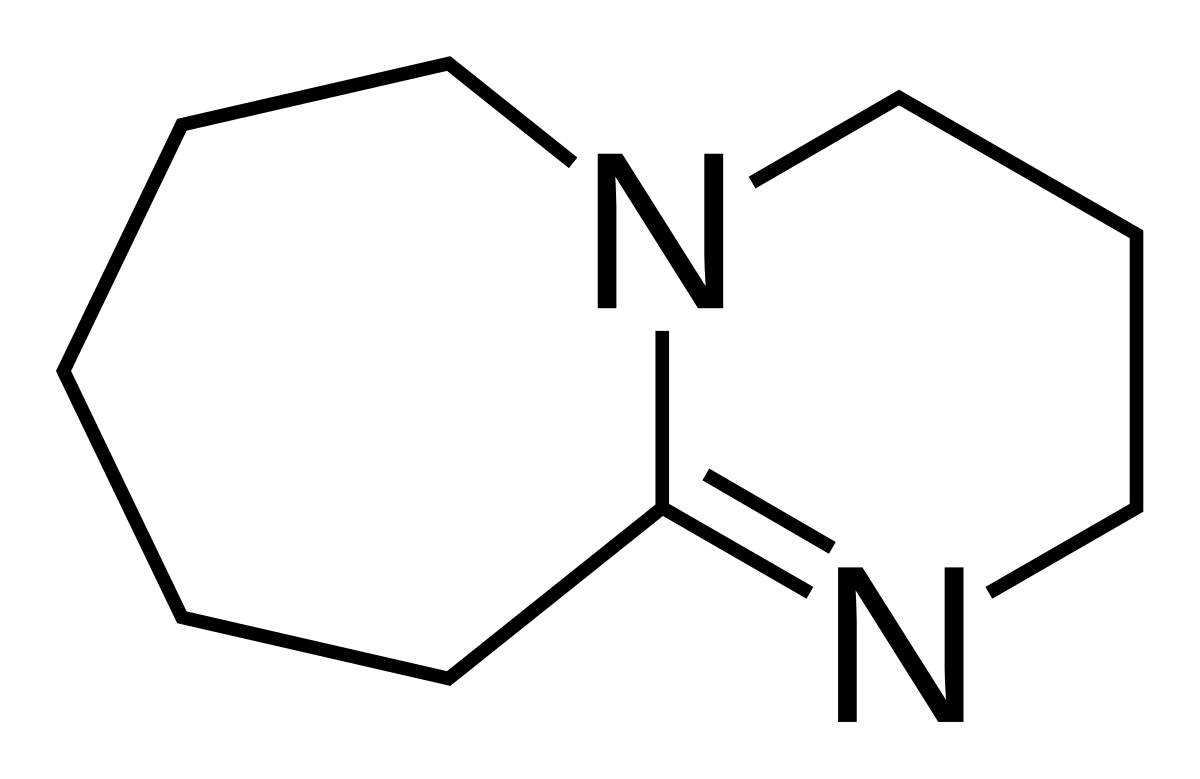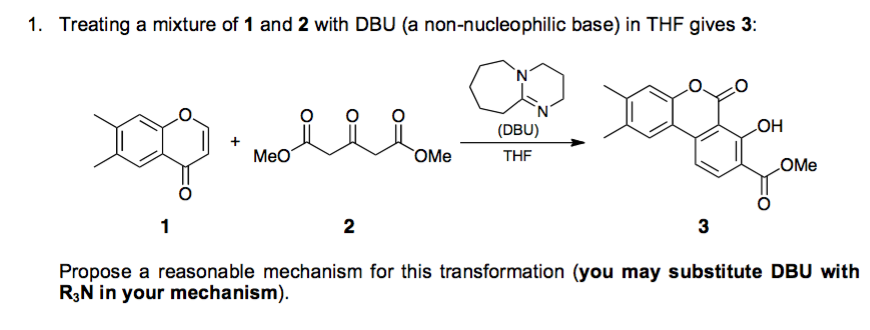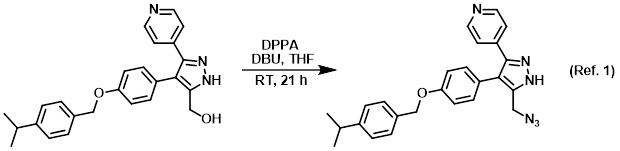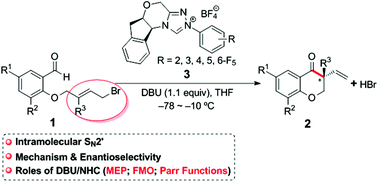
Theoretical study on the mechanism and enantioselectivity of NHC-catalyzed intramolecular SN2′ nucleophilic substitution: what are the roles of NHC and DBU? - Organic Chemistry Frontiers (RSC Publishing)

Proposed mechanisms for the DBU catalyzed urea-synthesis reaction from... | Download Scientific Diagram
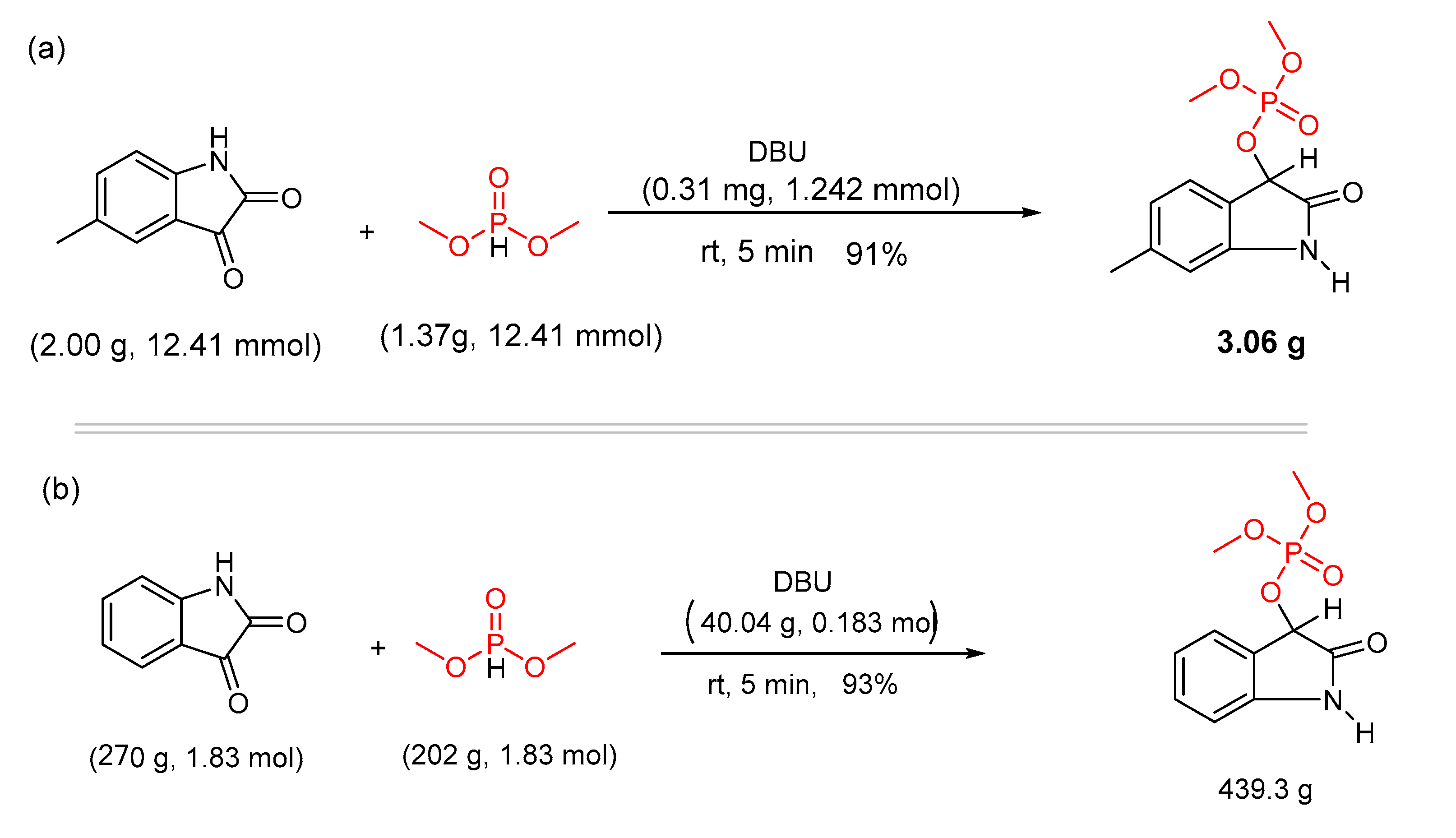
Catalysts | Free Full-Text | DBU Catalyzed Phospho-Aldol-Brook Rearrangement for Rapid Preparation of α-Phosphates Amide in Solvent-Free Conditions
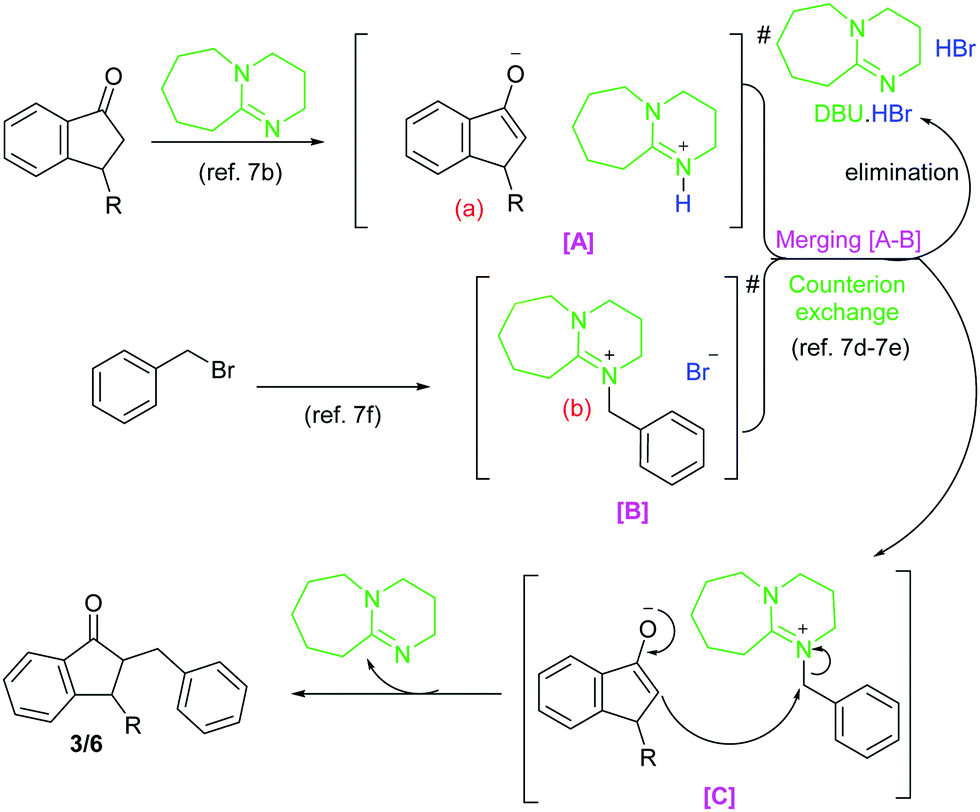
An efficient merging of DBU/enolate and DBU/benzyl bromide organocycles for the synthesis of alpha benzylated 1-indanone derivatives - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D2NJ00783E
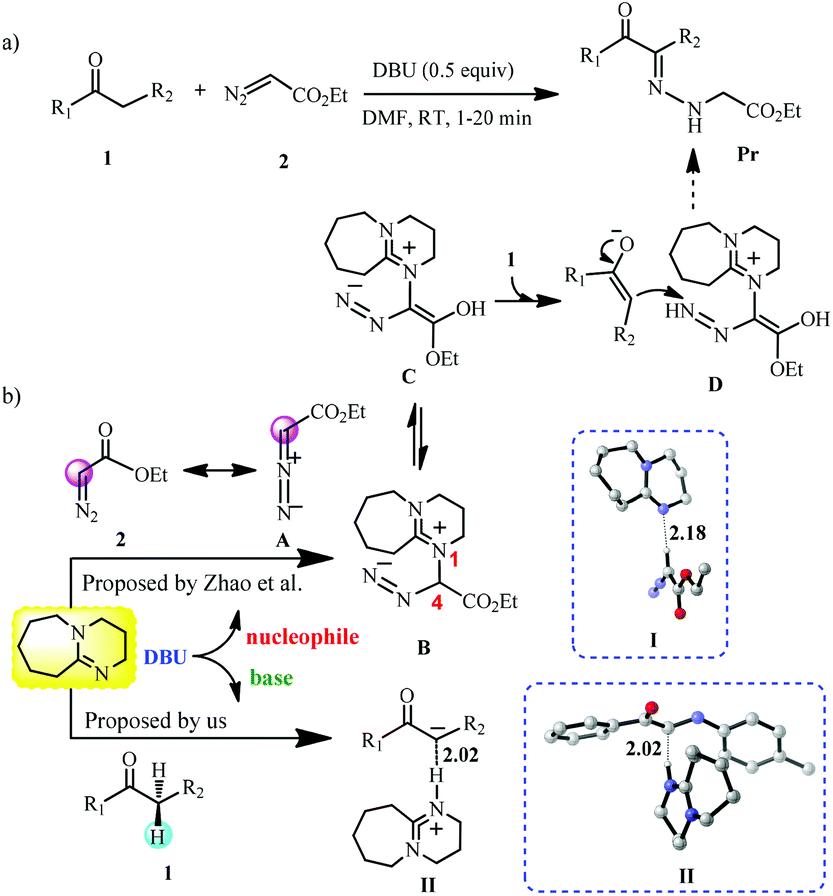
Mechanistic investigation-inspired activation mode of DBU and the function of the α-diazo group in the reaction of the α-amino ketone compound and EDA ... - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QO00602H
![Controlled Reactivity of 1,8‐Diazabicyclo[5.4.0]undec‐7‐ene (DBU) in the Selective Synthesis of 1‐(Bromoethynyl)arenes - Krishna Moodapelly - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library Controlled Reactivity of 1,8‐Diazabicyclo[5.4.0]undec‐7‐ene (DBU) in the Selective Synthesis of 1‐(Bromoethynyl)arenes - Krishna Moodapelly - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/da071cf8-eee0-4e2f-8e22-5b6f6a0f91e7/adsc201601279-fig-5004-m.jpg)
Controlled Reactivity of 1,8‐Diazabicyclo[5.4.0]undec‐7‐ene (DBU) in the Selective Synthesis of 1‐(Bromoethynyl)arenes - Krishna Moodapelly - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library

SciELO - Brasil - Mechanistic Investigation of DBU-Based Ionic Liquids for Aza-Michael Reaction: Mass Spectrometry and DFT Studies of Catalyst Role Mechanistic Investigation of DBU-Based Ionic Liquids for Aza-Michael Reaction: Mass Spectrometry
Proposed mechanisms for the DBU catalyzed urea-synthesis reaction from... | Download Scientific Diagram
![Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences](https://royalsocietypublishing.org/cms/asset/0ea92727-dfe5-4dad-b4fd-c68f2d8d3d5e/rspa20190238f03.jpg)
Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences
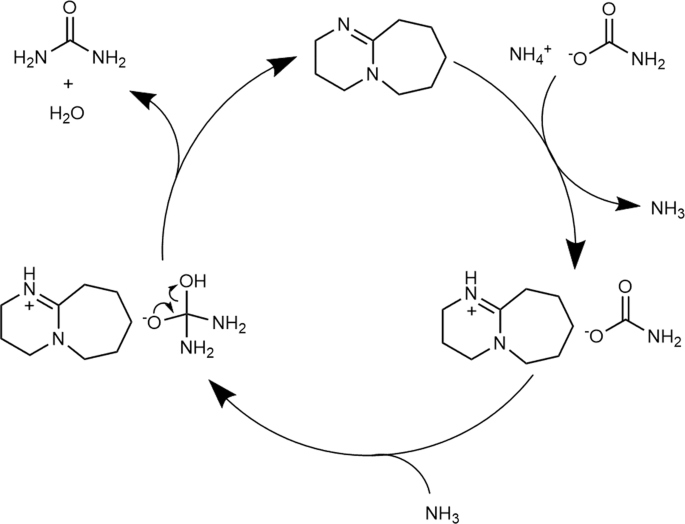
Organic bases catalyze the synthesis of urea from ammonium salts derived from recovered environmental ammonia | Scientific Reports
![College: Organic Chemistry] Does this mechanism make sense, the last step is confirmed in a paper but i'm trying to figure out the DBU catalysed mechanism (first step) : r/chemistryhomework College: Organic Chemistry] Does this mechanism make sense, the last step is confirmed in a paper but i'm trying to figure out the DBU catalysed mechanism (first step) : r/chemistryhomework](https://preview.redd.it/t5hrthkdks821.jpg?auto=webp&s=968746e147a1ecdbc999c52eeaa264aaa7ee2cc5)
College: Organic Chemistry] Does this mechanism make sense, the last step is confirmed in a paper but i'm trying to figure out the DBU catalysed mechanism (first step) : r/chemistryhomework
![Figure 1 from 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) and microwave-accelerated green chemistry in methylation of phenols, indoles, and benzimidazoles with dimethyl carbonate. | Semantic Scholar Figure 1 from 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) and microwave-accelerated green chemistry in methylation of phenols, indoles, and benzimidazoles with dimethyl carbonate. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0d4133dd6314ab3a6af35d9a5101b828f28a7059/2-Figure1-1.png)
Figure 1 from 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) and microwave-accelerated green chemistry in methylation of phenols, indoles, and benzimidazoles with dimethyl carbonate. | Semantic Scholar

Catalysts | Free Full-Text | DBU Catalyzed Phospho-Aldol-Brook Rearrangement for Rapid Preparation of α-Phosphates Amide in Solvent-Free Conditions
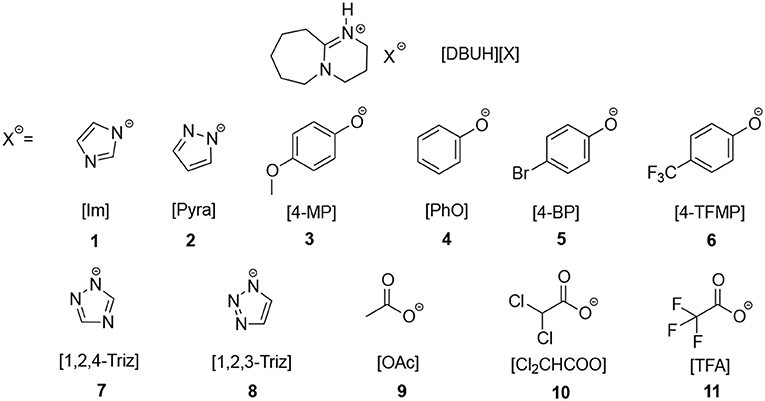
Frontiers | CO2 Absorption by DBU-Based Protic Ionic Liquids: Basicity of Anion Dictates the Absorption Capacity and Mechanism
![Synthesis of Azobenzenes Using N-Chlorosuccinimide and 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) | The Journal of Organic Chemistry Synthesis of Azobenzenes Using N-Chlorosuccinimide and 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.7b01530/asset/images/large/jo-2017-01530d_0002.jpeg)