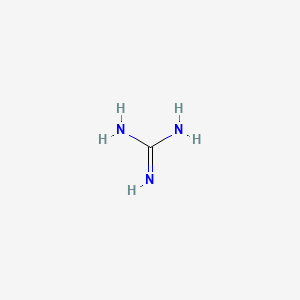
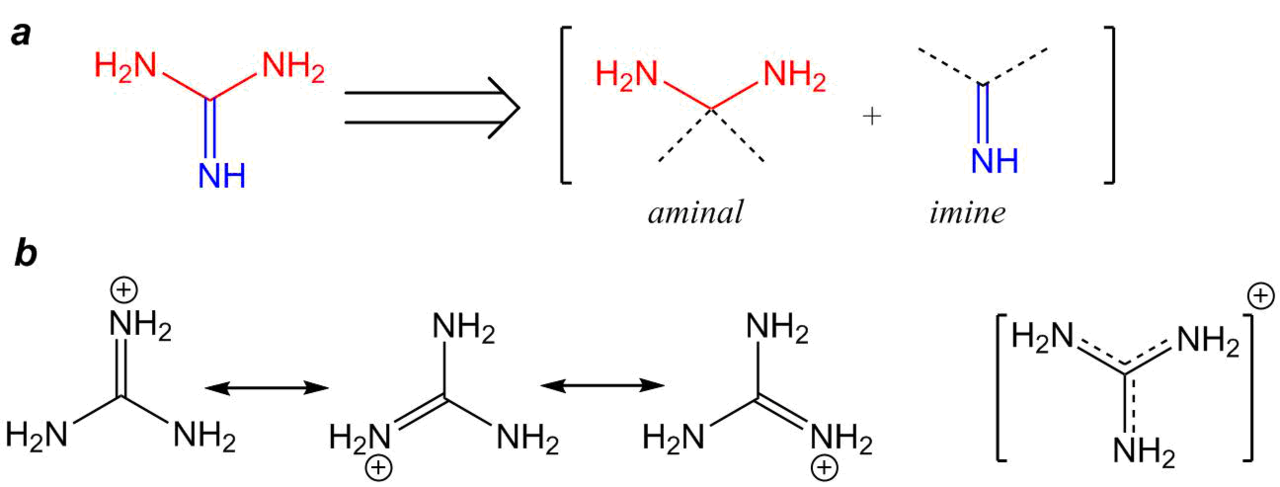
Guanidine and the guanidino group present in arginine are two of the strongest organic bases known. Account for their basicity. | Homework.Study.com
SOLVED: Guanidine is the strongest base among neutral organic compounds. The reason for the greater basicity of guanidine is: A. presence of three nitrogen atoms in the compound B. The delocalization of

Sequential Reduction of Nitroalkanes Mediated by CS2 and Amidine/Guanidine Bases: A Controllable Nef Reaction | Organic Letters
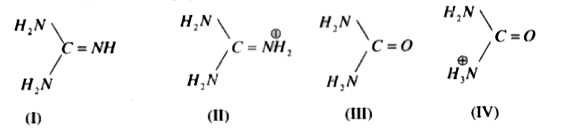
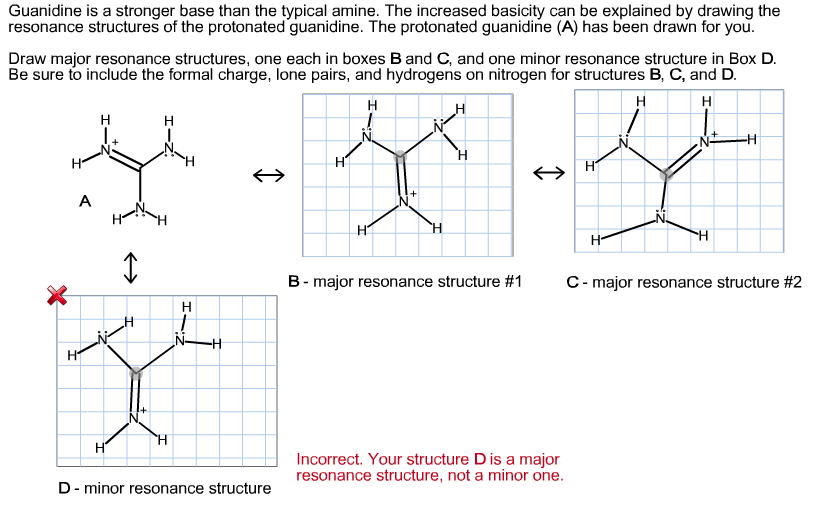
Guanidine (I) and its conjugate acid (II) are given below along with urea(III) and its conjugate base (IV) Basic properties of I & II compounds are mainly influenced by resonance and the
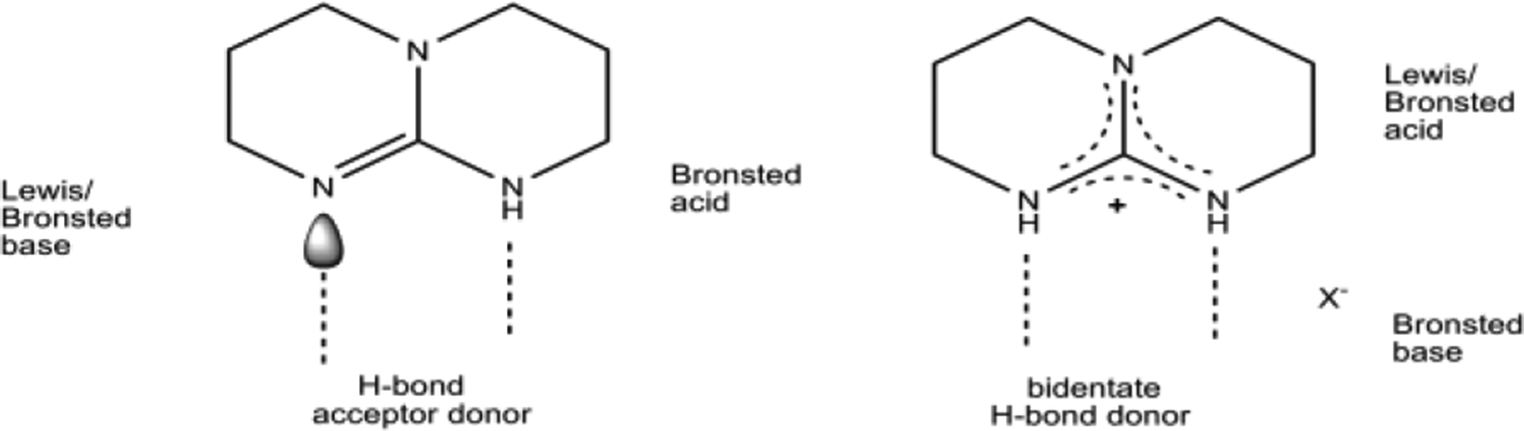
Recent Advances in Guanidine-Based Organocatalysts in Stereoselective Organic Transformation Reactions | IntechOpen

Strong Bicyclic Guanidine Base-Promoted Wittig and Horner−Wadsworth−Emmons Reactions | Organic Letters

A guanidine based bis Schiff base chemosensor for colorimetric detection of Hg(II) and fluorescent detection of Zn(II) ions - ScienceDirect
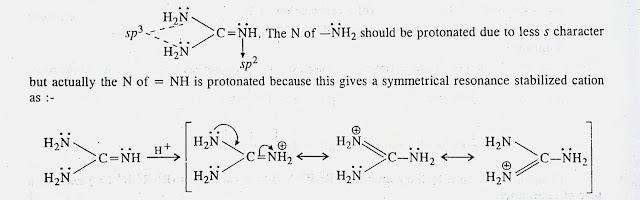
Welcome to Chem Zipper.com......: Give an explanation for the fact that Guanidine NH=C(CH3)2 is a stronger base than most of amines?