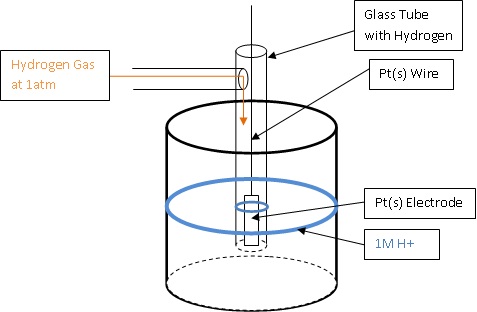
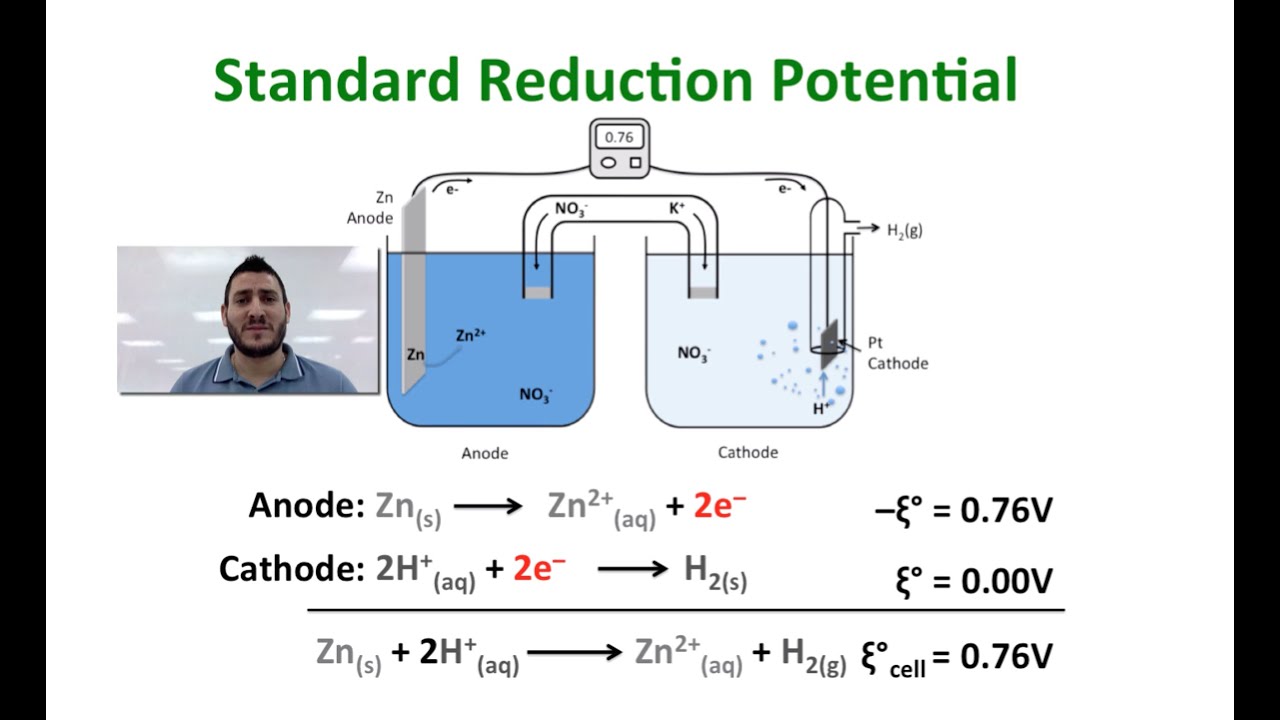
Calculation of Standard Reduction Potentials of Amino Acid Radicals and the Effects of Water and Incorporation into Peptides. | Semantic Scholar
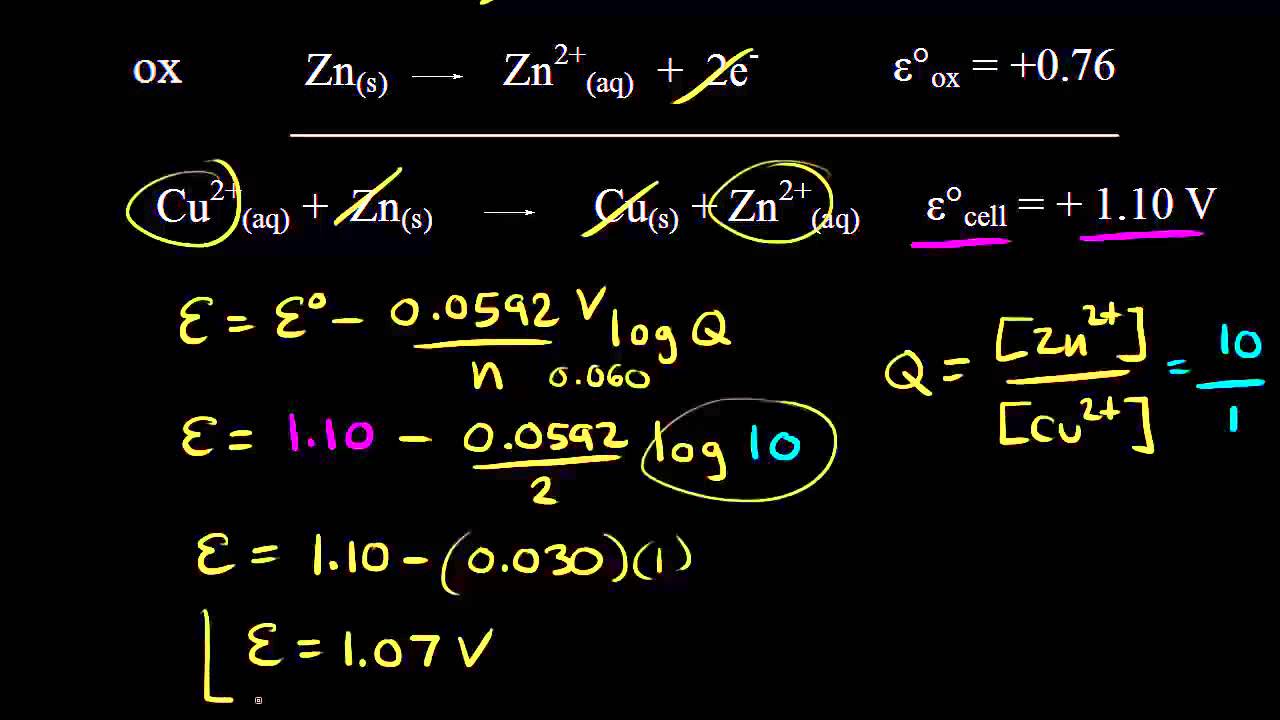
Using the Nernst equation | Redox reactions and electrochemistry | Chemistry | Khan Academy - YouTube

Question Video: Calculating a Cell Potential from Standard Electrode Potentials of Cadmium and Nickel | Nagwa

Standard Reduction Potentials for Oxygen and Carbon Dioxide Couples in Acetonitrile and N,N-Dimethylformamide | Inorganic Chemistry

The standard reduction potential for `Cu^(2+)|Cu` is `+0.34V`. Calculate the reduction potential... - YouTube

Question Video: Calculating the Standard Cell Potential for a Magnesium/Silver Galvanic Cell | Nagwa

:max_bytes(150000):strip_icc()/Standardreductionpotential-5b551731c9e77c003ec223b3.jpg)