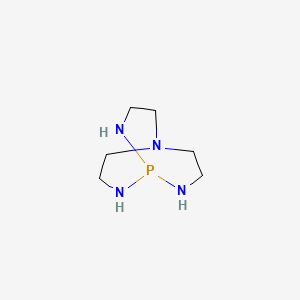
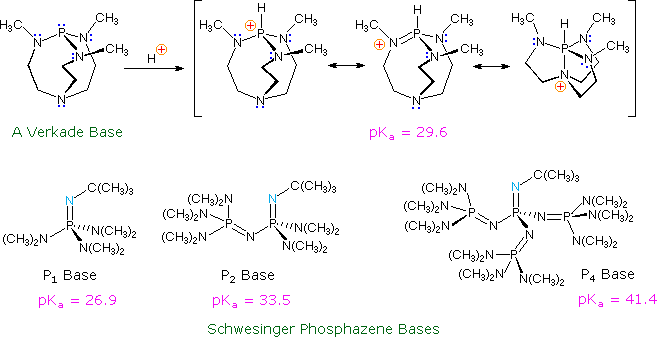
Verkade Base in FLP Chemistry–From Stoichiometric C–H Bond Cleavage to the Catalytic Dimerization of Alkynes | Organometallics

Two Verkade's super bases (Vkd_Me and Vkd_iPr) and two guanidine bases... | Download Scientific Diagram
![120666-13-9 | 2,8,9-Trimethyl-2,5,8,9-tetraaza-1-phosphabicyclo[3.3.3]undecane | Proazaphosphatrane; Verkade base | C9H21N4P | TRC 120666-13-9 | 2,8,9-Trimethyl-2,5,8,9-tetraaza-1-phosphabicyclo[3.3.3]undecane | Proazaphosphatrane; Verkade base | C9H21N4P | TRC](https://www.trc-canada.com/prod-img/T219435.png)
120666-13-9 | 2,8,9-Trimethyl-2,5,8,9-tetraaza-1-phosphabicyclo[3.3.3]undecane | Proazaphosphatrane; Verkade base | C9H21N4P | TRC

Verkade Base in FLP Chemistry–From Stoichiometric C–H Bond Cleavage to the Catalytic Dimerization of Alkynes | Organometallics

Interactions of Verkade's Superbase with Strong Lewis Acids: From Labile Mono- and Binuclear Lewis Acid–Base Complexes to Phosphenium Cations | Inorganic Chemistry
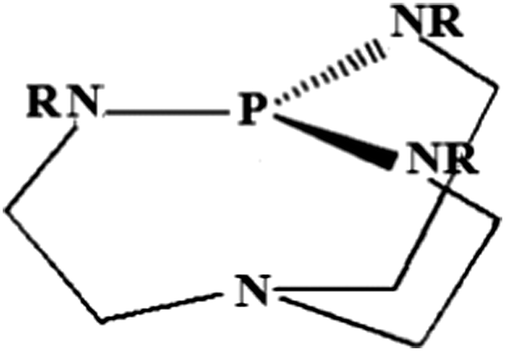
Protonation of Verkade bases: a theoretical study - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ04977G
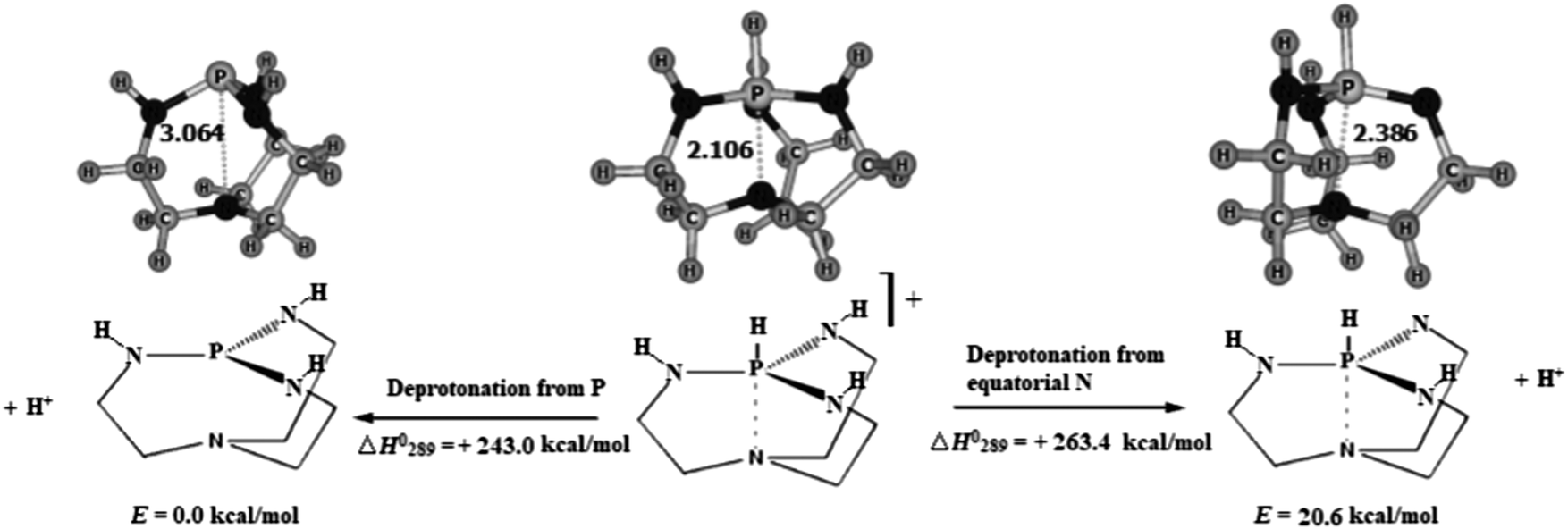
Protonation of Verkade bases: a theoretical study - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ04977G
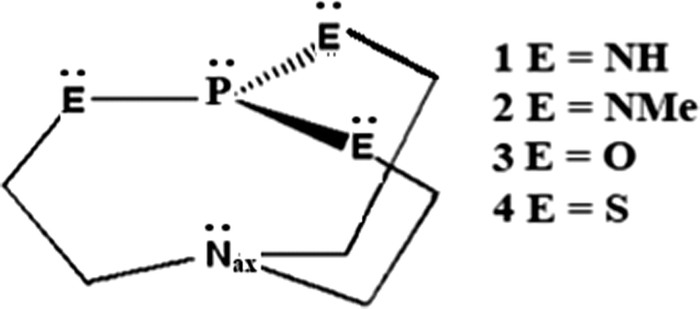
Protonation of Verkade bases: a theoretical study - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ04977G

Interactions of Verkade's Superbase with Strong Lewis Acids: From Labile Mono- and Binuclear Lewis Acid–Base Complexes to Phosphenium Cations | Inorganic Chemistry

Schematic representation of the considered phosphazenes and Verkade's... | Download Scientific Diagram

Verkade Base in FLP Chemistry–From Stoichiometric C–H Bond Cleavage to the Catalytic Dimerization of Alkynes | Organometallics

Verkade Base in FLP Chemistry–From Stoichiometric C–H Bond Cleavage to the Catalytic Dimerization of Alkynes | Organometallics

Interactions of Verkade's Superbase with Strong Lewis Acids: From Labile Mono- and Binuclear Lewis Acid–Base Complexes to Phosphenium Cations | Inorganic Chemistry

Interactions of Verkade's Superbase with Strong Lewis Acids: From Labile Mono- and Binuclear Lewis Acid–Base Complexes to Phosphenium Cations | Inorganic Chemistry

Interactions of Verkade's Superbase with Strong Lewis Acids: From Labile Mono- and Binuclear Lewis Acid–Base Complexes to Phosphenium Cations | Inorganic Chemistry

Verkade super base-catalysed transesterification of propylene carbonate with methanol to co-produce dimethyl carbonate and propylene glycol - ScienceDirect

Verkade Base in FLP Chemistry–From Stoichiometric C–H Bond Cleavage to the Catalytic Dimerization of Alkynes | Organometallics

Interactions of Verkade's Superbase with Strong Lewis Acids: From Labile Mono- and Binuclear Lewis Acid–Base Complexes to Phosphenium Cations | Inorganic Chemistry

Verkade Base in FLP Chemistry–From Stoichiometric C–H Bond Cleavage to the Catalytic Dimerization of Alkynes | Organometallics

Verkade Base in FLP Chemistry–From Stoichiometric C–H Bond Cleavage to the Catalytic Dimerization of Alkynes | Organometallics